MENU
Abstract-Pathogenesis and Diagnosis
Introduction
Etiology
Pathogenesis: Disc Associated Wobblers Syndrome
Pathogenesis: Bone Associated Wobblers Syndrome
Diagnosis: Clinical Signs
Diagnosis: Survey Radiographs
Diagnosis: Advanced Diagnostic Imaging
Diagnosis: Myelography and CT-Myelography
Diagnosis: Static Lesion versus Dynamic Lesion
Diagnosis: Magnetic Resonance Imaging
Conclusion
Bibliography
Abstract – Pathogenesis and Diagnosis
Wobbler syndrome in dogs refers to a disorder of the cervical vertebrae and intervertebral discs resulting in cervical canal stenosis and spinal cord compression. The main causes of wobbler syndrome are disc associated wobbler syndrome (DAWS) and bone associated wobbler syndrome (BAWS). Clinical signs in DAWS and BAWS are similar but the pathogenesis and the breeds affected are different. Magnetic resonance imaging is the diagnostic test to best define the site, severity, and nature of spinal cord compression, and allows visualization and characterization of lesions within the spinal cord. An early and accurate diagnosis is the critical factor for selecting the best treatment option and to improve the outcome of this devastating disease.
Introduction
“Wobbler syndrome” is the term most commonly used to refer to a canine disorder of the cervical vertebrae and intervertebral discs resulting in cervical canal stenosis and spinal cord compression. This term doesn’t indicate a specific diagnosis, but as in Horner’s syndrome indicates that the patient is affected by a specific set of abnormalities, mainly characterized by a “wobbling gait.” This wobbling gait is where the name originated. The most common underlying cause of “wobbler syndrome” is cervical spondylomyelopathy (CSM); however, “wobbler syndrome” can also be secondary to other diseases, such as infectious or inflammatory, developmental, acquired (e.g. subarachnoid cyst, syringomyelia), or neoplastic diseases of cervical spine.
Cervical spondylomyelopathy may be secondary to vertebral abnormalities or chronic intervertebral disc disease. When CSM is secondary to intervertebral discs diseases, it is called disc associated CSM or disc associated wobbler syndrome (DAWS). When CSM is secondary to vertebral abnormalities, it is called bone associated CSM or bone associated wobbler syndrome (BAWS).
Etiology
Disc associated wobbler syndrome tends to affect middle aged to older, large-breed dogs, with the Doberman Pinschers being over represented.1 However, many other breeds (including small dogs), have been reported to be affected by this condition. In one study of 90 dogs, where Doberman Pinschers were excluded, thirty-two breeds were represented. Among these, Labrador Retrievers (13), Dalmatians (13) and Rottweilers (12) were the most commonly affected.2 Doberman Pinschers may be predisposed to clinically significant cervical spinal cord compression by relative congenital vertebral canal stenosis, with a loss of reserve space.1 The main factor in DAWS, however, is the underlying chronic degenerative disc disease. The C5-C6 and C6-C7 disc spaces are most commonly affected, with lesions in both interspaces present in about 20% of affected dogs.3
Bone associated wobbler syndrome is typically a disease of young adult giant breed dogs, mainly Great Danes and Mastiffs. Cranial cervical vertebrae are more commonly affected and multiple spaces tend to be involved. The main factor in BAWS is a combination of vertebral malformations and osteoarthritic changes of the articular facets.1
Pathogenesis
Disc Associated Wobbler Syndrome
Disc associated wobbler syndrome in dogs shares many similarities with the cervical spondylotic myelopathy in people, and the Doberman breed has been proposed as a natural model to study the disease in humans.4,5 Although the pathogenesis of DAWS is not well understood, it is thought to be multi-factorial, and includes primary developmental abnormalities and secondary degenerative changes that lead to vertebral canal stenosis and spinal cord compression. Spinal cord compression in DAWS is typically secondary to a combination of the protrusion of the intervertebral disc and the hyperthrophy or “in-folding” of the ligamentous structures surrounding the spinal cord (dorsal longitudinal ligament, dorsal annulus, interarcuate ligament and joint capsules). The spinal cord compression is often dynamic, in that the amount of compression changes with changes in spinal position (i.e. elevated vs. flexed vs. turned laterally). It could be that the increased thickness of the surrounding ligament structures is compensatory to an underlying vertebral instability. (Fig. 1)
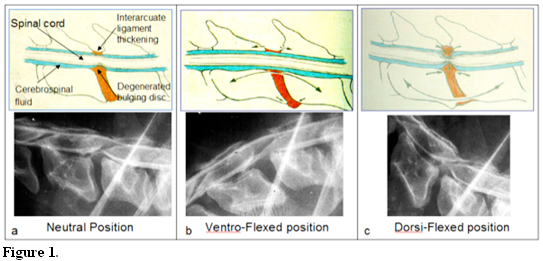
Schematic representation (upper row) and related radiographic abnormalities (lower row) showing the dynamic nature of DAWS. The upper row shows some of the anatomical structures involved in the of the spinal cord compression. The lower row is a myelography of a Doberman affected by DAWS. The degree of the spinal cord compression (a) is alleviated by placing the neck in a ventro-flexed position (b) and increased by placing the neck in an extended position (c). The dorsi-flexed view is not longer recommended because it may exacerbate the neurologic signs. The dynamic nature of DAWS syndrome explains why dogs affected by this disease are reluctant to raise their neck.
However, instability, which has been defined as the loss of ability of the cervical spine under physiologic loads to maintain its normal pattern of displacement so that there is no damage to the spinal cord or nerve roots, has not yet been documented in dogs.4,5 The collapsed disc space may also cause narrowing of the intervertebral foramina, which in turn may lead to nerve entrapment and spinal artery compression with subsequent radicular pain and decreased blood supply to the spinal cord.6-8 Generally, these are traction-responsive lesions, in which the degree of spinal cord compression may be reduced by the application of traction to the cervical spine. The dynamic nature of the spinal cord compression is one of the key factors in establishing the diagnosis of DAWS. However, interpretation of the traction-responsiveness of lesions is highly subjective and dependent on personal opinion.9,10 Furthermore, MRI has revealed that almost any compressive lesion, whether dynamic or static, will improve with traction.9 Application of cervical spinal traction re-establishes disc width, flattens the redundant soft tissue structures, and opens the narrowed foramina. In most dogs, 2-3 mm of distraction restores a normal disc width of 4-6 mm.3
A somewhat common event in DAWS is the occurrence of a “domino lesion,” also called “adjacent segment disease,” which is the development of a second lesion at an adjacent disc space after surgical treatment. (Fig. 2)
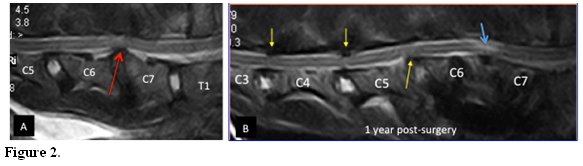
Domino lesions. MRI (T2-weighted sagittal) of a 7 year-old Doberman showing a DWAS at C6-C7 (red arrow). (A) One year post-surgery, (B) The dog was treated with distraction fusion using a cortical bone graft and stabilized with titanium screws and PMMA. The dog presented one year later with chronic progressive tetraparesis. New compressive lesions, at C5-C6 ventrally, and at C4-C5 and C3-C4 dorsally (yellow arrows) were evident and likely as consequence of “domino effect”. A new area of signal hyperintensity is also evident at the treated site (blue arrow). Courtesy of Dr. G. Destrero
It is unclear whether the development of this additional lesion is the natural progression of an underlying similar process at the adjacent disc space, or if it is an accelerated degenerative process influenced by the biomechanical effect of fusion at the treated site.11 The influence of the latter is supported by the documentation, on a long term follow-up, of a higher rate of adjacent-segment disease in children treated with anterior cervical fusion for fracture and dislocation, and in patients with Klippel-Feil syndrome, who have congenital cervical fusion of the cervical vertebrae.11,12 Fig. 2a
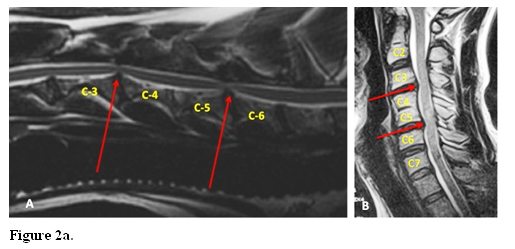
Klippel-Feil syndrome “like” lesion. Sagittal MRI T2 weighted of a 9 year-old mix-breed dog showing contiguity of the vertebral body of C4-C5 and cervical compression at the adjacent C3-C4 and C5-C6 sites (A). This lesion shows similarly with Klippel-Feil Syndrome in people as seen in B. Sagittal T2 MRI T2 weighted of cervical spine of a human affected by Klippel-Feil syndrome showing vertebral fusion of the vertebral body of C3 and C4 and spinal compression at both adjacent sites (B).
These stresses can exacerbate any pre-existing subclinical in- stability and produce either disc extrusion or hypertrophy of annular or ligamentous structures.13,14 A domino lesion, with recurrence of paraparesis to tetraparesis, develops in up to one-third of dogs after ventral decompression or metal implant insertion and bone cement fixation.3,14 This is typically reported between 6 months and 4 years after the original surgery, with a mean time to recurrence of approximately 2 years. 10,16,17
Bone Associated Wobbler Syndrome
The pathogenesis of BAWS is different. Because the disease is seen predominantly at an earlier age, it is thought to be a congenital or developmental abnormality of the cervical vertebrae (particularly in the Great Dane), (Fig. 3a) or secondary to degenerative joint disease of the cervical articular facets and pedicle (particularly in the Mastiff), resulting in progressive dorsal and/or lateral spinal cord compression. (Fig 3b)
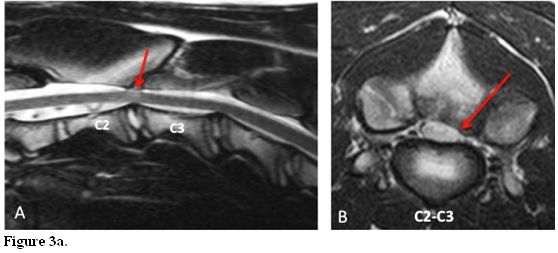
MRI (T2 weighted) images of a 5 month old Great Dane affected by BAWS. Sagittal image shows normal disc at C2-C3with appropriate signal intensity. (A) Transverse section at C2-3 shows severe extradural dorso-lateral spinal cord compression and distortion of the spinal cord. The compression is secondary to dorsal arch deformity with articular facets joint degenerative changes (red arrows) with vertebral canal stenosis consistent with BAWS.(B)
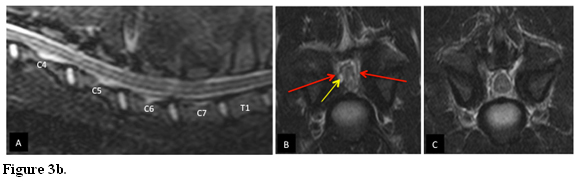
MRI (T2 weighted) images of a 12 month old Mastiff affected by BAWS. Sagittal image shows normal discs with appropriate signal intensity. (A) Transverse section at C6-7 shows severe extradural bilateral spinal cord compression and distortion of the spinal cord. The compression is secondary to articular facets joint degenerative changes with bony proliferation (red arrows) and synovial cyst (yellow arrow) with vertebral canal stenosis is consistent with BAWS.(B) Transverse section at the adjacent cranial disc space (C5-C6) shows a less severe degenerative facets joints changes without spinal cord compression.(C)
At surgery and post-mortem examination of dogs in various study, incomplete cartilage coverage and asymmetry of the articular process was found.18 These malformations can result in altered mechanical stresses on the joint cartilage, with subsequent degenerative joint disease and ankylosis.18 Extradural synovial cyst may also be present (more often in Mastiff) as a complication of the degenerative arthritic facet changes, leading to unilateral or bilateral lateral spinal cord compression. BAWS is a usually a static compression and typically the intervertebral discs are not affected.1,5
Diagnosis
Clinical Signs
Dogs with DAWS are usually 4-8 years of age, while dogs with BAWS are usually younger. Animals affected with DAWS and BAWS have similar clinical signs, and a chronic progressive history (weeks to months) is the typical presentation. Clinical signs may range from only cervical pain (5-10% of patients) to acute non-ambulatory tetraparesis (10% of patients).1 Paralysis is usually the acute decompensation of a chronic lesion. Although cervical pain is not commonly part of the presenting complaint, upon careful neurologic exam it is observed in approximately 65% to 70% of Dobermans and 40-50% of other breeds.1 Decreased cervical range-of-motion, most commonly in dorsi-flexion and lateral flexion, is also typical and it can be evaluated using either gentle manual manipulation or food treat.1 Deep palpation of the transverse process may also elicit pain. Elbow abduction with internal rotation of the digits (“toe-in” posture) may also be seen in about one-third of Dobermans with DAWS.1 Proprioceptive deficits may be seen in all four legs, or may be limited only to the hind legs. The lateral distribution of the afferent proprioceptive pathway related to the hind legs, in contrast to the more medial distribution of the afferent pathway of the front legs, explains why the hind legs are more often affected.17,19 However, in chronic cases, despite the ataxic gait, proprioceptive deficits may not be detected. This can be explained by the fact that different tracts carry the pathways for conscious and unconscious proprioception.1
The most common presentation of DAWS is ataxia, which is more severe in the pelvic limbs, and low carriage of the head. A broad-based stance may be noticed in the hind limbs when the dog is standing. Affected dogs often show a characteristic gait, with the thoracic limbs moving with short stilted steps and the pelvic limbs moving with wide elongated steps. (Fig. 4)
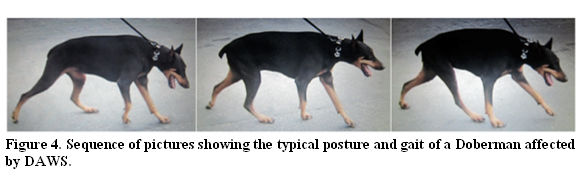
The neck is typically hold either along an horizontal plane or ventro-flexed, the hind limbs are hold wide apart and move with long steps, while the front limbs move with short steps with various degree of stumbling.
This is also known as a “disconnected” or “2-engine” gait, where the thoracic limbs seem to advance at a different rate. Scuffing of the pelvic or thoracic limb toes/nails also can be seen. The owners commonly report a gradual onset, although the symptoms can sometimes occur or become exacerbated acutely.1 This disease is usually progressive if left untreated and the prognosis is usually worse for dogs with chronic clinical signs and those with non-ambulatory tetraparesis or tetraplegia.
Survey Radiographs
Survey radiographs may be suggestive of DAWS but they are not conclusive. They are used as a preliminary screening to rule out other potential causes of cervical diseases, such as vertebral fractures, subluxations, or tumors, and bone or disc infectious diseases. Survey radiographs in dogs with DAWS may reveal narrowing of the intervertebral disc space, mild deformity of the cervical vertebral body (i.e. rhomboidal shape, vertebral tipping) and spondylosis deformans ventral to the intervertebral space. However, changes on survey radiographs do not always correlate with spinal cord compression and they may be normal in some dogs with DAWS. It has been reported that approximately 20-25% of clinically normal Dobermans have radiographic changes comparable to those seen in dogs with DAWS.1 Cervical vertebral ratios may be useful as a screening test for dogs with DAWS.20 Osteoarthrosis and sclerosis of the articular facets at the affected sites may be seen in dogs with BAWS.1
Advanced Diagnostic Imaging
Diagnosis of DAWS can be made by myelography, myelography and computed tomography (CT), or Magnetic resonance imaging (MRI).
Myelography and CT-Myelography
Myelography alone or in combination with CT (CT-myelography), may be considered an option if MRI is not available. (Fig. 5)

Severe narrowing and wedging of the intervertebral disc space between C6-C7 is present; malformation of the cranioventral border of C7 is also evident. Severe ventral spinal cord compression is noted (a). The severity of the compressive lesion reduces remarkably in size with traction (b). Myelography and Computed Tomography (c). Transverse section of a spinal cord during Myelography and Computed Tomography at the affected site. The contrast injected in the space around the spinal cord (myelography: circular white line pointed by the red arrows), shows the distorted spinal cord shape (a normal spinal cord shape has a round circular shape) secondary to by ventral bulging disc and its associated ligamentous structures (asterisk).
CT-myelography allows visualization on cross sectional views of the spinal cord compression and spinal cord atrophy. Spinal cord atrophy may be seen as a triangular shape to the spinal cord and a widening of the subarachnoid space around the spinal cord.21 As previously described with DAWS, the extent of spinal cord compression can be dynamic and vary with flexion, extension, and linear traction (distraction) of the cervical spine. However, due to the risk of neurologic deterioration from cervical manipulation during general anesthesia, only linear traction myelography or linear traction MRI views continue to be routinely used.1 Comparison of a neutral view with a linear traction view is used to distinguish static versus dynamic compression.3,22
Static Lesion versus Dynamic Lesion
The concept of static and dynamic lesions was first established in 1982.4 The determination of the “dynamic” or “traction responsive” feature, in contrast to “static” lesions (such as extruded disc material, malformed facets, or deformed vertebral arches), has been suggested as being essential for the diagnosis of DAWS.22 However, guidelines for performance and interpretation of linear traction views are lacking. The way the traction is applied, the amount and the degree of traction, has never been standardized except for two recent studies, which proposed the use of a cervical harness and traction forces not greater than 20% and 25% of the dog’s weight, respectively.9,23 This should generate enough traction force to alleviate traction-responsive lesion causing the spinal cord compression. (Fig. 6)
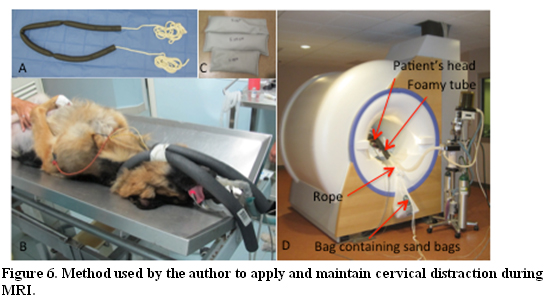
In a foamy soft hallowed tube it is inserted a long rope, leaving the free ends untied.(A) The tube is positioned around the dog neck in a U shape and the two arms of the tube are fixed close to each other with a regular bandage tape at the level of the dog chin. This apparatus can be placed prior to move the patient into the MRI, as shown in the picture, (B) or can be applied once the patient is inside the MRI. Once inside the MRI, the front legs are pulled caudally and are tight at the caudal end of the MRI table. To generate the amount of traction required (between 20% and 25% of the dog’s weight), a number of sand bags of different weigh can be used in combination and placed in a single bag.(C) Just prior to perform the traction MRI, the bag containing the sand bags is tight to the free ends of the rope, and after apply a manual gentle traction of the rope attached to the dog neck, the bag with its weight is left hanging out of the MRI gantry.(D)
However, distinction between dynamic and static lesions is very subjective and difficult to determine.1 Furthermore, a comparative myelographic and MRI study showed that lesions that were considered dynamic on myelography appear static on MRI, and that any compressive lesion either on myelogram or MRI may improve with traction.9 One major disadvantage of myelography is the potential occurrence of post-myelographic seizures and a temporary worsening of the patient’s neurological status.24-26
Magnetic Resonance Imaging
MRI is the best and least invasive way to diagnose DAWS and BAWS in dogs. MRI best defines the site, severity, and nature of spinal cord compression, and allows visualization and characterization of lesions within the spinal cord.9,27 (Fig. 7 and 8)
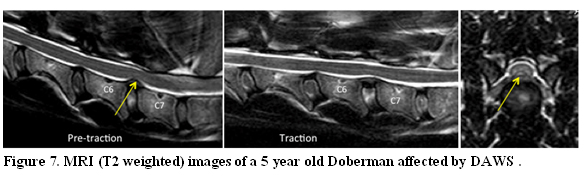
Pre-traction sagittal image shows a ventral extradural compression at C6-C7 (arrow). (A) Sagittal image during traction shows improvement in the degree of ventral compression.(C) This lesion is considered dynamic. Transverse section pre-traction at the affected site shows the bulging disc (arrow) causing ventral midline spinal cord distortion.(C)
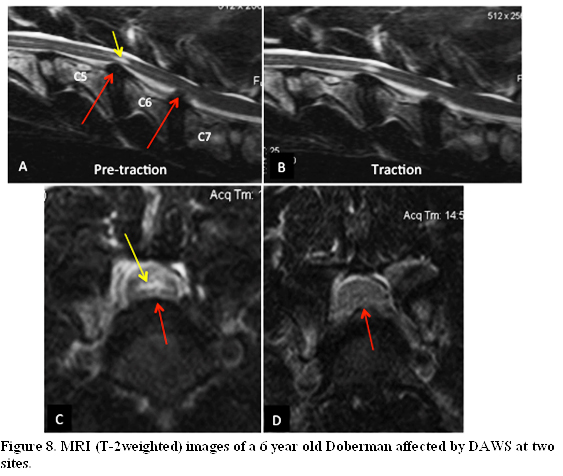
The dog presented with two-year history of ataxia-tetraparesis treated with activity restriction and non-steroidal anti-inflammatory therapy, and with 3 days history of acute tetraplegia. On sagittal pre-traction images the spinal cord is ventrally compressed at C5-C6 and C6-7 (red arrows); both discs at these sites appear to be degenerated, an intraspinal area of hyperiintensity (yellow arrow) is visible at C5-C6 and indicative a more chronic lesion at this site. (A) Traction sagittal images reveal minimal improvement in the C5-C6. and moderate improvement in the C6-C7 spinal cord compression. (B). This lesion could be considered dynamic. Transverse T2W image at the C5-C6 intervertebral space shows moderate ventral spinal cord compression (blue arrow) with central cord hyperintensity (yellow arrow). (C) Transverse T2W image at the C6-C7 intervertebral space shows severe ventral spinal cord compression, with loss of the normal CSF around the cord, which was most likely responsible for the acute and rapid progressive tetraplegia. (D)
Intraparenchymal signal changes are seen in approximately 50% of dogs with DAWS, and provide identification of acute versus chronic lesions.1 This may be particularly useful not only in establishing severity of the lesion, but also chronology when multiple areas are affected. Hyperintensity on T2-weighted images (T2) usually indicates chronicity and more severe damage to the spinal cord. It is not uncommon in the same patient to detect two or more sites of spinal cord compression and only in one of them see signal hyperintensity on T2. This may be interpreted as the evolution of the same problem at different stages at different sites. Association between spinal cord MRI findings and histologic abnormalities has been well documented in people, and the type of intra-spinal cord abnormalities seen on MRI may be of prognostic value. Hyperintensity on T2 combined with isointensity on T1-weighted images (T1) is usually associated with mild histologic abnormalities, mainly characterized by mild loss of neuronal cells, gliosis and edema in the gray matter, as well as demyelination, edema and Wallerian degeneration in the white matter. In contrast, hyperintensity of T2 combined with hypointensity on T1 are usually associated with more severe histologic changes, mainly characterized by necrosis in the gray and white matter.28 Hyperintensity on T2 doesn’t appear to be correlated with prognosis in dogs, but if it is combined with hypointensity on T1 it may be associated with a worse prognosis.29 In DAWS, T2 hypointensity in the disc space reflect dehydration of the nucleus pulposus and disc degeneration, typically associated with various degree of disc protrusion and spinal cord compression. Intervertebral foraminal stenosis may also be seen, and it may play an important role in spinal pain. Interestingly, in some dogs the degree of spinal cord compression may be minimal relative to the severity of the clinical signs. In these patients, the dynamic factor may play an essential role in development of the clinical signs.1,29 In BAWS, osseous proliferation from degenerated articular facets, dorsal laminae and pedicles may be visible as hypointense changes on T1 and T2 and the spinal cord compression is best visible on transverse and dorsal views. Synovial cysts may be seen adjacent to the articular facets as a homogeneous area of hyperintensity on T2.1,24 A dynamic study with linear traction can be performed using the same guidelines described for myelography.9,24
Conclusions
The main causes of wobbler syndrome in dogs are DAWS and BAWS. Although the clinical signs are similar in both conditions, the pathogenesis and the breeds affected are different. Magnetic resonance imaging is the diagnostic test to best define these conditions. The MRI visualization and characterization of the lesions within the spinal cord may also be of prognostic value. An early and accurate diagnosis is the critical factor for selecting the best treatment option and to improve the outcome of this devastating disease.
Bibliography
1. da Costa RC. Cervical Spondylomyelopathy (Wobbler syndrome). In R. da Costa, eds.Veterinary Clinics of North America, Small Animal Practice, Spinal Diseases. Philadelphia, PA: Elsevier. 2010; Vol. 40, pp. 881-913.
2. Jefferey N, McKee. W. Surgery for disc associaye wobbler syndrome in the dog – an examination of the controversy. J Small Anim Pract. 2001; 42, 574-581.
3. Trotter E. Cervical spine locking plate fixation for treatment of cervical spondylotic myelopathy in large breeds. Vet Surg 2009; 38, 705-718.
4. da Costa R. 2012. Comparison of cervical spondylomyelopathy in dogs and cervical spondylomyelopathy in people. In Proceeding. Annu Meet Am Coll Vet Int Med Forum; New Orleans.
5. Sharp NJ, Cofone M, Robertson ID, et al. Cervical spondylomyelopathy in the Doberman dog: a potential model for cervical spondylotic myelopathy in humans. J Invest Surg 1989;2:333.
6. Panjabi MM, et al. Cervical Spine Kinematics and Clinical Instability. The Cervical Spine Research Society. The Cervical Spine4th 2005:55-78.
7. Sharp N, Wheeler S. Cervical disc disease. In: Sharp W, ed. Small animal spinal disorders. 2nd ed. Edinburgh: Elsevier Mos- by, 2005;93–120.
8. Morio Y, Teshima R, Nagashima H, et al. Correlation between operative outcomes of cervical compression myelopathy and MRI of the spinal cord. Spine 2001;26:1238–1245.
9. Lemarie RJ, Kervin S, Partington B, et al. Vertebral subluxation following ventral cervical decompression in the dog. J Am Anim Hosp Assoc 2000;36:348–358.
10. da Costa RC, Parent JP, Dobson H, et al. Comparison of magnetic resonance imaging and myelography in 18 Doberman pinscher dogs with cervical spondyl-omyelopathy. Vet Radiol Ultrasound 2006;47(6):523–31.
11. Rusbridge C, Wheeler S, Torrington A, et al. Comparison of two surgical techniques for the management of cervical spondylomyelopathy in Dobermans. J Small Anim Pract 1998;39:425–431.
12. McGrory B, Klassen R. 1994. Arthrodesis of the cervical spine for fractures and dislocations in children and adolescents. A long term follow-up study. J Bone Joint Surg Am:1606-1614.
13. Guille JT, Miller A, Bowen JR, et al. 1995. The natural history of Klippel-Foil syndrome: clinical, roentgenographic, and magnetic resonance imaging findings at adulthood. J Pediatr Orthop:617-626.
14. Bruecker K, Seim H, Blass C. Caudal cervical spondylomyelopathy: de- compression by linear traction and stabilization with Steinmann pins and polymethyl methacrylate. J Am Anim Hosp Assoc 1989;25:677–683
15. Sharp N, Wheeler S. Cervical spondylomelopathy. In: Wheeler S, eds. Small animal spinal disorders, diagnosis and surgery. 2nd ed. Edinburgh: Elsevier Mosby, 2005;211–246.
16. Downes C, Gemmill T, Pink J, et al. Traction-responsive cervical disc lesions in 90 dogs, in Proceedings Br Small Anim Vet Cong 2009. Available at: www.vin.com/Members/Proceedings/Proceedings.plx? CID=bsava2009&PID=pr32447&O=VIN. Accessed Apr 7, 2010.
17. Bruecker KA, Seim HB 3rd, Withrow SJ. Clinical evaluation of three surgical methods for treatment of caudal cervical spondylomyelopathy of dogs. Vet Surg 1989;18:197–203.
18. Trotter EJ. Canine Wobbler Syndrome. In Newton CD and Nunamaker DM, Texbook of Small Animal Orthopaedics. Eds. JB Lippincott Company, Philadelphia PA,, 1985;765-784.
19. King AS. In Phisiological and Clinical Anatomy of the Domestic Mammals. Vol 1, Central Nervous Syste. Eds Oxford Science Publications. 1994, 86-99.
20. da Costa RC, Johnson JA, Parent JM. Are cervical vertebral ratios useful in the diagnosis of cervical spondylomyelopathy in Dobermans? ACVIM Forum Proceedings. Lakewood (CO): American College of Veterinary Internal Medi- cine; 2010. p. 332.
21. Sharp NJ, Cofone M, Robertson ID, et al. Computed tomography in the evaluation of caudal cervical spondylomyelopathy of the Doberman Pinscher. Vet Radiol Ultrasound 1995;36(2):100–8.
22. Seim HB, Withrow SJ. Pathophysiology and diagnosis of caudal cervical spondylomyelopathy with emphasis on the Doberman Pinscher. J Am Anim Hosp Assoc 1982;18:241–51.
23. Penderis J. Use of traction during magnetic resonance imaging of caudal cervical spondylomyelopathy (“wobbler syndrome”) in the dog. Vet Radiol Ultrasound 2004;45:216–219.
24. Barone G, Ziemer LS, Shofer FS, et al. Risk factors associated with development of seizures after use of iohexol for myelography in dogs: 182 cases (1998). J Am Vet Med Assoc 2002;220(10):1499–502.
25. Lewis DD, Hosgood G. Complications associated with the use of iohexol for myelography of the cervical vertebral column in dogs: 66 cases (1988-1990). J Am Vet Med Assoc 1992;200(9):1381–4.
26. da Costa RC, Parent JM, Dobson H. Incidence and risk factors of postmyelo- graphic seizures in dogs: 503 cases. J Vet Intern Med 2009;23(3):707–8.
27. Lipsitz D, Levitski RE, Chauvet AE, et al. Magnetic resonance imaging features of cervical stenotic myelopathy in 21 dogs. Vet Radiol Ultrasound 2001;42(1):20–7.
28. Ohshio I , Hatayama A , Kaneda K, et al. Correlation between histopathologic features and magnetic resonance images of spinal cord lesions. Spine 1993;18(9):1140–9.
29. da Costa RC, Parent JM. One-year clinical and magnetic resonance imaging follow-up of Doberman Pinschers with cervical spondylomyelopathy treated medically or surgically. J Am Vet Med Assoc 2007;231(2):243–50.
